Metabolic changes allow eggs to store fuel for development
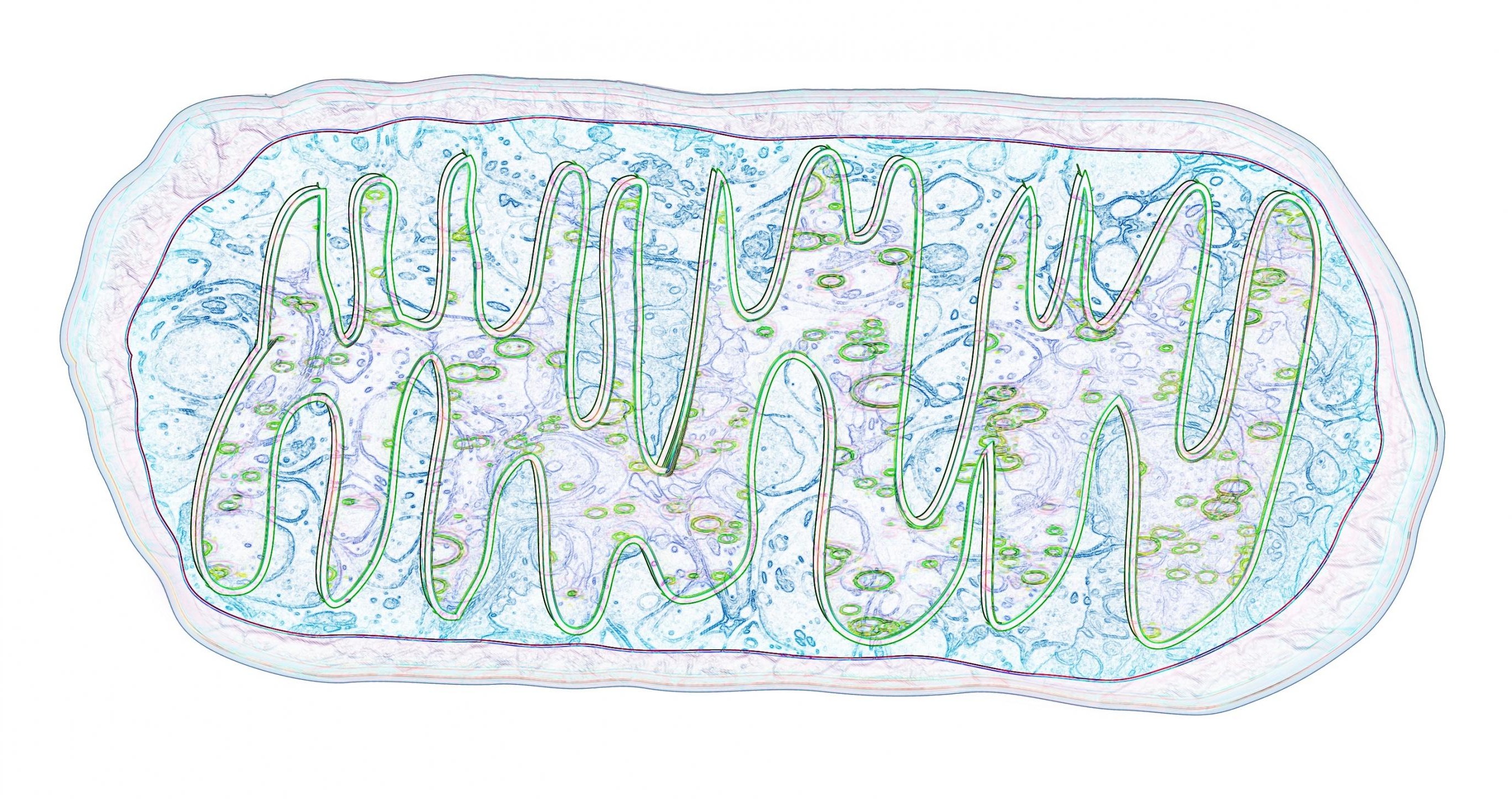
This is an artist’s depiction of mitochondria, courtesy of Matthew Sieber. The purple on the outside border represents the outer mitochondrial membrane. The blue on the inside depicts the mitochondrial inner membrane space. The green and purple inside depicts the inner mitochondrial membrane and mitochondrial matrix. Image credit: Matthew Sieber.
Long before fertilization, an egg lays the groundwork for its future. In addition to providing half the DNA required for a complete genome, the egg must also store nutrients essential for early development as a fertilized embryo. Without proper fuel, its future is uncertain at best.
Changes in metabolism – the chemical reactions that convert food into energy, create the building blocks of life, and handle waste – are surely key to the egg’s ability to stockpile nutrients instead of breaking them up for immediate use. But exactly when and how the egg does this has been something of a mystery.
Solving this mystery – how eggs store fuel for development – is the focus of JCC Fellow Matthew Sieber’s work at the Carnegie Institution. And his recent results reveal important molecular links between infertility and metabolic diseases like obesity and diabetes.
Previously, studies on nutrient storage within the egg, also called an oocyte, have been limited, according to Sieber, who completed the work with Michael Thomsen and HHMI Investigator Allan Spradling.
“It is very difficult to obtain cells at specific stages of development for metabolic analysis,” says Sieber. Plus, most research on metabolic changes during development has been done in somewhat artificial cell culture systems.
“To address these concerns, we came up with a paradigm to isolate pure populations of developing oocytes at different stages of development from in vivo tissue,” he says. “Once we had pure populations of the cell, we had to adapt current methods and technologies to work on very small sample sizes (due to limited material) to perform in-depth metabolomic, transcriptomic, and proteomic analysis.”
Using fruit fly egg cells, Sieber and his colleagues focused on mitochondria, a cellular structure that converts nutrients into energy. Experiments showed that, at the end of egg development, insulin signaling is inactivated, and sugars accumulate – sugars that are later used as fuel for the developing embryo.
How does the cell make that shift? The egg cells enter a low activity state called quiescence, where lowered insulin signaling triggers the enzyme GSK3 to remodel the electron transport chain (ETC). The ETC normally converts sugars into usable energy, but when remodeled by GSK3, sugars are stockpiled for later.
The results, which were published recently in Cell, are not limited to fruit flies. “Intriguingly, we observed similar ETC remodeling and glycogen uptake in maturing Xenopus oocytes, suggesting that these processes are evolutionarily conserved aspects of oocyte development,” Sieber says, referring to similar results he found in a frog species. “This work allowed us to generate a very clear picture of how metabolism is changing during oocyte production and provides a model to understand the mechanisms that link nutrition to reproductive disorders.”
Allan Spradling adds, “We believe our findings are directly applicable to understanding human infertility. Polycystic ovary syndrome is the leading cause of female infertility and it is strongly associated with diabetes and insulin resistance, which suggests that defects in the metabolic aspects of oocyte maturation may be the underlying source of the disorder.”
Additionally, understanding how cells enter and exit a state of metabolic quiescence could have far reaching future implications and therapeutic applications. “The aberrant exit from quiescence by stem cells could underlie aspects of cancer initiation,” says Sieber. “Moreover, the inability to exit quiescence during aging may contribute to the decline in the wound healing response during aging.”
For now, Sieber aims to understand the mechanisms that mediate mitochondrial quiescence and how quiescence is reversed after fertilization. “I’m also very interested in how disrupting metabolic mechanisms during germline development may influence the resulting progeny, and I’m currently investigating how disruptions of metabolism in the germline may influence embryogenesis and progeny susceptibility to metabolic syndromes.”
Sieber recently won the Carnegie Postdoctoral Innovation and Excellence Award and will soon enter the job market, seeking a faculty position at a research institution. He’s grateful for the support of the JCC Fund. “Particularly, I would like to thank current and former Board of Scientific Advisors such as Anjana Rao, Carol Prives, Haifan Lin, and Rich Losick, as well as many others, for their helpful suggestions and mentorship while conducting this work and developing my independent research program.”