Dr. Carla Kim joins the Board of Scientific Advisors
Carla Kim, Ph.D., has joined the Board of Scientific Advisors (BSA) for the Jane Coffin Childs Fund for Medical Research. Dr. Kim is a Principal Investigator at Boston Children’s Hospital and Professor of Genetics and Pediatrics at Harvard Medical School. Research in Kim’s lab is broadly interested in the role of stem cells in normal lung development, and in lung diseases including lung cancers.

Dr. Carla Kim
Scientific Training
An interest in gene therapy drew Kim to graduate school. She knew she didn’t want to be a medical doctor, but was interested in understanding diseases and dissecting their underlying mechanisms.
As a doctoral student in Dr. John Petrini’s lab at the University of Wisconsin, Kim’s research focused on a protein complex, or cellular machine, that is involved in DNA repair. DNA repair is a process where a cell “fixes” damage to its DNA like how spell-check works for written documents. Quite a bit was known about this complex in budding yeast. However, the study of this complex in mammals was quite a challenge. For example, deleting the mouse gene encoding Rad50, one of the proteins that makes up this cellular machine, renders mice nonviable, making it difficult to study the biological impact of this complex.
Drawing on what was known about the yeast Rad50 protein, Kim cleverly engineered a more subtle mouse model. Instead of deleting the Rad50 gene entirely, Kim included Rad50 with one single amino acid change which had previously been identified to disrupt Rad50’s function in yeast. This strategy proved to be more tractable, as some of these mice survived through birth. Interestingly, one of two things happened to these mice. Some died young due to dysfunction in their blood stem cells, whereas others lived longer and were prone to develop cancer. Kim’s studies were an important technical breakthrough and provided key insights into the Rad50 DNA repair complex’s function in mammals.
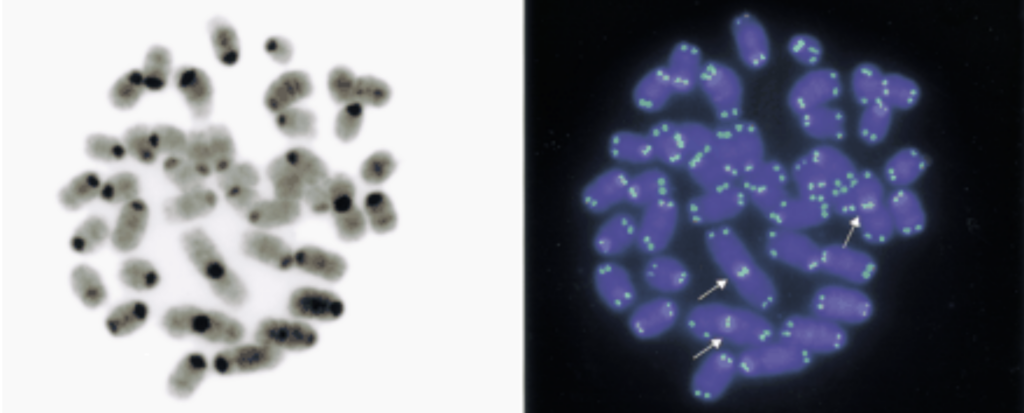
Chromosomal instability in lymphoma cells from Kim’s Rad50 mouse model (Bender et al, 2002)
Yet, Kim was most interested in the potential connection her model revealed between stem cells and cancer. At the time adult stem cells and their roles in cancer were being investigated in breast and blood cancers, but were unknown for most solid organs.
JCC Fellowship
For her postdoctoral training Kim joined Dr. Tyler Jacks’ lab. Jacks and his lab are experts at developing novel mouse models for many types of human cancers. Kim encourages burgeoning scientists to “go where the training is the best”, and views her postdoc move as having done just that.
Erica Jackson, a graduate student in Jacks’ lab, had just developed a mouse model of lung cancer using a mutated form of the K-ras protein that is found in many human cancers. Additionally, just before starting her postdoc, Kim attended a stem cell conference with a memorable poster presentation by Barry Stripp, Ph.D. Stripp had used his own mouse model of lung injury to identify a cell population that starts dividing after the injury. Kim realized that Stripp’s data was strong evidence for the presence of adult stem cells in the lung, and that Jackson’s mouse model was an ideal system for investigating the connection between lung stem cells and lung cancer.
During Kim’s postdoc she applied for and was awarded a JCC Fellowship supporting her research. In her JCC application Kim laid out the ambitious scope for her postdoctoral research:
The goal of this research proposal is to identify and characterize the primary target cell population in lung adenocarcinoma, the most predominant form of lung cancer in the United States.
Kim adapted techniques she had learned in her doctoral research to isolate and characterize bronchioalveolar stem cells from the lungs of normal mice. These stem cells reside in the space where small breathing tubes meet the air sacs in the lungs. This is part of Kim’s prescription for success: find the intersection between new ideas and your previous knowledge and then use that knowledge to make headway into the unknown.
Kim next showed that there were more bronchioalveolar stem cells in the mice with lung cancer, suggesting that the stem cells might assist cancer growth. Indeed, Kim used a chemical stimulant to show that activating bronchioalveolar stem cells also enhanced lung cancer initiation and growth.
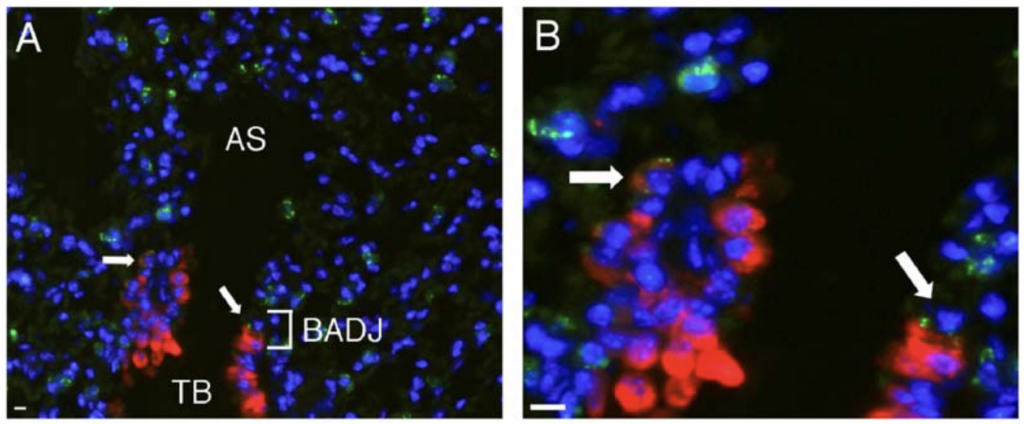
Arrows point to bronchioalveolar stem cells in normal lung (Kim et al, 2005).
In short, during her postdoc Kim had achieved her lofty goal and identified the primary target cell population in a common form of lung cancer. Yet, in her JCC research proposal Kim explained how this achievement would be only the tip of the iceberg in terms of understanding the role of stem cells in normal lung biology and in different lung diseases:
Identification of lung adenocarcinoma cells of origin will provide a basis for novel methods of early tumor detection and intervention. … Finally, the identification and characterization of lung stem cells will be important for innovative approaches to treatment of not only cancer, but also a variety of lung diseases.
Kim Lab
After launching her own lab, Dr. Kim and her team have continued to map out fundamental lung cell biology and reveal the cellular foundations of various lung diseases, including lung cancer. For example, they identified cancer stem cell populations in two most common subtypes of human lung cancer: lung adenocarcinomas with differing genotypes and lung squamous cell carcinoma.
An especially enabling innovation was their development of a three-dimensional lung organoid system. Traditional two-dimensional cell culture grows cells – lung cells or any other cell type – flat on a dish. This flat setup poorly mimics the textured environments within which cells reside in a real organism. As such, cells often lose key characteristics that are important for their function when they’re cultured in 2D. Growing cells in 3D organoids often better retains these key characteristics. Such was the case with Kim’s 3D lung organoids which they used to differentiate lung stem cells into specialized adult lung cells. This organoid system provides a better approximation of the cellular diversity present in lungs, and powered the study of interactions between different lung cell types.
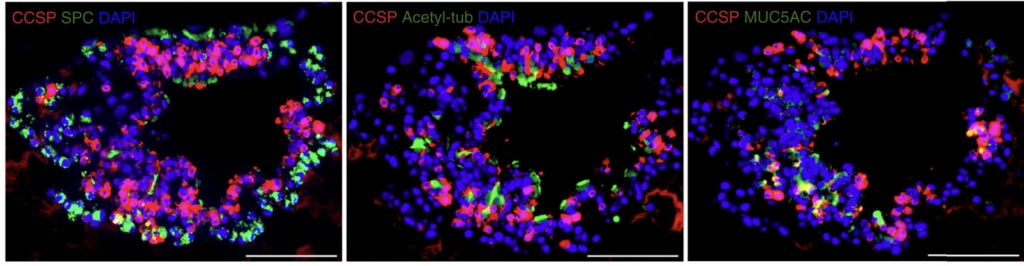
Images of lung organoids developed by Kim’s lab (Lee et al, 2014).
Kim and her lab have also applied their abundant lung cell biology knowledge and innovative toolkit to diseases and procedures beyond lung cancer including bronchopulmonary dysplasia, obliterative bronchiolitis, cystic fibrosis, and lung transplantation.
Giving back to JCC as an Advisor
Reflecting on her own JCC Fellowship, Kim recalls the boost of confidence and feeling of validation for her scientific ideas. Furthermore, Kim notes that it was especially sweet to win a fellowship after having missed out on others.
As a JCC Scientific Advisor, Kim looks forward to supporting Fellows who are doing the most exciting research and fostering the careers of early investigators. Kim advises JCC Fellows to follow what they’re most passionate about in their research, and to make sure that they are in the right environment with the best support to help launch their independent careers. Welcome back to JCC, Dr. Kim!