The Jane Coffin Childs Fund Welcomes Dr. Kathryn Wellen to its Board of Scientific Advisors
Kathryn Wellen Ph.D. has joined the Jane Coffin Childs Fund for Medical Research Board of Scientific Advisors. Dr. Wellen is a Professor and Vice Chair of the Department of Cancer Biology at the University of Pennsylvania. The Wellen Lab is focused on metabolism and metabolic regulation of cell identity and function, including in cancer cells.

Kathryn Wellen Ph.D.
Scientific Background
Wellen discovered her passion for scientific research as an undergraduate in Dr. Lizabeth Allison’s lab at the College of William and Mary. Her foray into research was studying the cellular localization of the thyroid hormone receptor in frogs. During this experience, Wellen realized that she loved the hands-on puzzle solving aspect of research, as well as the thrill of uncovering something brand new – no matter how minute – that had never been known before.
Next, Wellen joined Dr. Gökhan Hotamişligil’s lab at Harvard University where she investigated the connection between metabolism and inflammation. It was known that overeating and chronic overexposure to nutrients trigger inflammation in diseases such as obesity and diabetes. Wellen hypothesized that, under healthy conditions, cells employ protective mechanisms that prevent normal levels of essential nutrients from inappropriately triggering inflammation.
To find the link between metabolism and inflammation, Wellen searched for genes that were differentially expressed due to inflammatory and metabolic triggers. Focusing on those that were also highly expressed in fat cells led Wellen to STAMP2 (six-transmembrane protein of prostate 2). She found that mice without STAMP2 developed inflammatory responses to nutrients in a normal diet and developed metabolic disease similar to as if they were eating high-fat diets. Wellen’s study revealed that STAMP2 is a novel integrator of inflammation and nutrient availability.
Metabolic Regulation of Gene Expression
You have evolved from worm to man, but much within you is still worm.
– Friedrich Nietzche
Biologically speaking Nietzche was on the right track, but we can actually trace many of our common molecular roots even further back than worms. A lot of what we know about gene expression, and most areas of cell biology for that matter, we learned from single-cell organisms like Baker’s yeast. One example of this is histone acetylation.
Histones are proteins that package our DNA such that some genes will be exposed and available to be expressed, whereas other genes that are not needed will be tightly packaged away. Acetylation is like a green light added to histone proteins that helps determine which genes should be open and expressed.
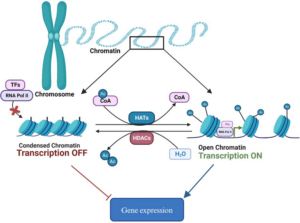
Role of histone acetylation in gene expression from Bajpai et al, 2024.
Acetyl-CoA is the proximal molecular building block that cells use to acetylate histones. In yeast cells acetyl-CoA is converted from acetate, which is enriched in their surrounding environment. While human cells can perform this same reaction, unlike yeast we’re not bathing in abundant pools of acetate that we can absorb and use to drive histone acetylation reactions.
Dr. Wellen’s postdoctoral research in Dr. Craig Thompson’s lab at the University of Pennsylvania discovered where the acetyl-CoA for mammalian histone acetylation comes from. Glucose is converted into acetyl-CoA, by the enzyme ATP-citrate lyase (ACL). Wellen convincingly exhibited that ACL plays a critical role in determining the extent of histone acetylation in several mammalian cell types. Zooming out, Wellen’s groundbreaking study indicated how the metabolic state of cells impacts gene expression to regulate cell identity and function.
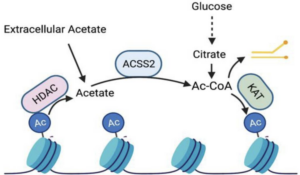
Model for Acetyl-CoA feeding into histone acetylation from Guertin & Wellen, 2023.
The Wellen Lab
Dr. Wellen established her independent lab in the Department of Cancer Biology at the University of Pennsylvania in 2011. The enzyme ATP-citrate lyase continues to be a central focus of Wellen’s research. By developing advanced tools to investigate the biology of this enzyme, and others, Wellen’s lab is at the cutting edge of demonstrating how metabolism changes in different cell types, both in healthy and disease states.
For example, Wellen’s lab is providing insight into how our bodies process increasingly sugar-laden diets. They’ve demonstrated how there are sex-specific differences in metabolizing high-sugar diets where ATP-citrate lyase in adipocytes promotes fat storage in females to a much greater extent than in males. Additionally, they’ve shown that fructose – the main sugar in high-fructose corn syrup – is processed by the gut microbiota which in turn supplies the liver with the resulting acetyl-CoA.
Wellen’s lab is also interested in how metabolism contributes to the development and progression of cancer. The big idea here is that cancer cells use different variations of metabolism compared to healthy cells, and by understanding these differences one could potentially find new drug targets or diet alterations that would synergize with existing cancer drugs. Her lab’s research includes exemplary examples of how understanding the mechanistic details of metabolism will help inform and comprehend such therapeutic approaches.
They’ve identified that ATP-citrate lyase is a logical drug target in prostate cancer, and one that would likely synergize with existing prostate cancer drugs. Building on her postdoctoral research, Wellen’s group demonstrated that acetyl-CoA reprograms glioblastoma cells to specifically promote the expression of genes related to cell adhesion and cell migration – two functions that contribute to cancer spreading throughout the body via metastasis. This is exciting because it shows that acetyl-CoA is not just activating the expression of genes at random. Rather, acetyl-CoA is like a switch that turns on existing gene regulatory programs which dictate cell identity and function.
Wellen’s lab has also explored other metabolic enzymes beyond ATP-citrate lyase. Recently they looked into a protein complex involved in breaking down the branched-chain amino acids leucine, isoleucine, and valine. Deleting one subunit of this complex, but only in pancreatic cells, accelerated the early stages of pancreatic cancer development. In contrast, inhibiting another subunit in tissues other than the pancreas, put the brakes on early stages of pancreatic cancer. This is a tremendous example of the same metabolic reaction promoting opposite functional outcomes in different cell types.
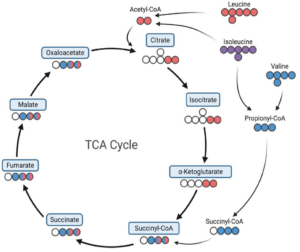
Metabolic processing of branched-chain amino acids from Noji et al, 2025.
Advice for Jane Coffin Childs Fellows
Dr. Wellen is excited to join the Jane Coffin Childs Fund Board of Scientific Advisors to help shape and mentor the next generation of breakthrough scientists. Wellen encourages JCC Fellows to continue thinking outside the box and asking new, transformative questions with their research. Furthermore, she advises Fellows to search out their own scientific niche, one that becomes synonymous with their name in the minds of colleagues and peers.
We welcome Dr. Wellen to the JCC Board of Scientific Advisors!