New Research from JCC Fellow Dr. Emily Rundlet
Jane Coffin Childs Fellow Emily Rundlet, Ph.D., has discovered an antigen for antibodies that neutralize monkeypox virus. Dr. Rundlet and her colleagues used a novel AI-based strategy to rapidly identify and prioritize potential antigens for experimental validation. In addition to this important monkeypox discovery, Rundlet’s approach serves as a roadmap that could accelerate vaccine development timelines during future outbreaks.

JCC Fellow Emily Rundlet, Ph.D.
The road back to virology
During her undergraduate studies Rundlet spent a semester at the NIH conducting HIV research in the lab of Peter Kwong, Ph.D. Her time in the Kwong lab was just after Jason McLellan, Ph.D., her current postdoc advisor, had left to start his own independent career.
After graduation, Rundlet took a hiatus from virology while working as a research technician at Caltech, and then as a graduate student in Scott Blanchard, Ph.D.’s lab at Weill Cornell Medicine. During her graduate studies, Rundlet developed her expertise in cryo-EM and made key structural insights into the process of protein translation.
For her postdoc Rundlet returned to virology ready to apply her structural biology skills to both fundamental and applied projects. McLellan’s lab was a perfect fit.
Discovering the monkeypox antigen
Out of the plethora of viral projects in McLellan’s lab, monkeypox captured Rundlet’s interest. Monkeypox doesn’t follow the typical entry and fusion rules of many other viruses, so there appeared to be rich and unexplored biology that Rundlet could uncover. Additionally, her collaborators in Emanuele Andreano Ph.D.’s lab had identified 12 neutralizing antibodies for monkeypox, yet the antigen(s) for these antibodies were still unknown.
That’s where Rundlet came into the project. Monkeypox has over 40 annotated surface-exposed proteins, so experimentally testing every possible combination of these proteins with the 12 neutralizing antibodies would have been slow and expensive. Instead, Rundlet cleverly applied AlphaFold 3 (AF3) to rapidly model 480 antigen-antibody pairs. The highest-scoring interactions involved two of the antibodies binding to the monkeypox protein OPG153 (Figure 1).
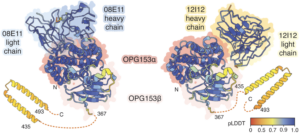
Figure 1. High confidence modeled interactions between two neutralizing antibodies and the monkeypox protein OPG153.
Rundlet and her coauthors quickly solved cryo-EM structures for each antibody bound to OPG153. These structures were similar to the modeled predictions, thereby validating her prediction process (Figure 2).
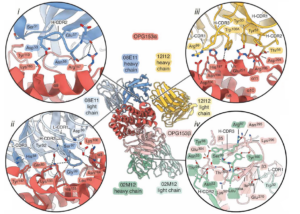
Figure 2. Cryo-EM structures of the interaction between two neutralizing antibodies and OPG153 are similar to the modeled predictions, while the interaction with a third antibody was not predicted.
Interestingly, when testing all of the 12 neutralizing antibodies for binding to OPG153 in a biophysical assay, Rundlet found that six additional antibodies also bound to this protein. This suggests that many of the lower-confidence predictions from her AF3 screen are also bona fide complexes.
Beyond revealing a monkeypox target for the majority of the collected neutralizing antibodies, two key takeaways from Rundlet’s research are:
- Predictive modeling can rapidly prioritize antigen targets for testing, which may be especially critical during global outbreaks.
- AI-assisted modeling of protein complexes is likely useful across broad swathes of biology. Rundlet’s approach serves as an early benchmark on how to apply this modeling.
Mining viruses for biological gems
Now that Rundlet has identified OPG153 as a key target for neutralizing antibodies, she next wants to figure out what exactly OPG153 does. The literature describing monkeypox biology is rather sparse, so Rundlet will work on deciphering why OPG153 is such a key target for neutralizing antibodies.
Long term, Rundlet would like to lead her own lab investigating translational regulation in host-virus interactions. She notes that “viruses hold a lot of these little gems”. Rundlet gets a thrill out of mining these gems to help design new medicines and aims to generate novel lab tools.